
PART.1 Preface
The urgent need to enhance the performance of lithium-ion batteries (LIBs) is becoming more and more prominent, especially in terms of ensuring reliable operation under extreme cold conditions. All-solid-state batteries (ASSBs) offer a promising solution to the challenges faced by conventional LIBs in low-temperature environments. In this study, we take advantage of amorphous solid-state electrolytes (SSEs) x Li₃N-TaCl₅ (1 ≤ 3x ≤ 2) to develop ASSBs that can operate efficiently under extremely low-temperature conditions. The designed ASSBs employ a LiCoO₂ cathode (with a mass loading of 4.46 mg cm-² ) and Li-In negative electrode with initial discharge capacities of 183.19, 164.8, and 143.78 mAh g-1 at -10, -30 °C, and -40 °C, respectively, at a current density of 18 mA g-¹, and at the 100th cycle exhibited a final discharge capacity of 137.6 mAh g-1 at -30 °C and 18 mA g-1. In addition, the initial discharge capacity of the ASSBs was 51.94 mAh g-¹ after cycling at -60°C for more than 200 hours at a current density of 18 mA g-¹.
PART.2 Graphical Quick Facts
Figure 1: Schematic of the structure of a customized ASSB for extreme low-temperature environments.
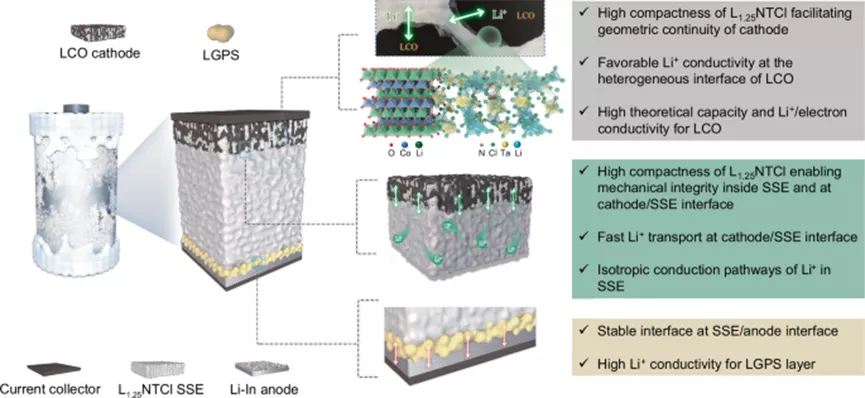
The ASSB is composed of LiCoO₂ (LCO) as the cathode, amorphous Li₁.₂₅N₀.₄₁₇-TaCl₅ (L₁.₂₅NTCl) as the solid-state electrolyte (SSE), Li₁₀GeP₂S₁₂ (LGPS) as the interface layer, and Li-In as the anode. This structural configuration ensures the mechanical and chemical stability of the battery, as well as favorable ion transport at the SSE and electrode/SSE interface, even under extremely low-temperature conditions.
Figure 2: Structural analysis of amorphous xLi₃N-TaCl₅.

a Schematic diagram of the combination of Li₃N and TaCl₅, used to form amorphous xLi₃N-TaCl₅ (left), and the structure and coordination polyhedra of the amorphous xLi₃N-TaCl₅ obtained through ab initio molecular dynamics simulation (right). b XRD patterns of amorphous xLi₃N-TaCl₅ prepared under different ball milling conditions for 30 hours (1 ≤ 3x ≤ 2). c XRD patterns of L1.25NTCl prepared under different ball milling conditions. d Raman spectra of TaCl₅ and amorphous L1.25NTCl. e Least squares refinement of the PDF data of amorphous L1.25NTCl using the small box method, and the local PDFs of Ta—Cl, Ta—N, Li—Cl, and Li—N pairs extracted from the total PDF data.
Figure 3: Properties of amorphous xLi₃N-TaCl₅.

The a-c Nyquist plots depict the impedance responses of the xLi₃N-TaCl₅ solid-state electrolyte under different 3x values (a), different ball-milling conditions (b), and different temperatures (c). (d) The ionic conductivity of amorphous xLi₃N-TaCl₅ (1 ≤ 3x ≤ 2) at 25 °C. e The ionic conductivity of L1.25NTCl prepared under different ball-milling conditions at 25 °C. f The Arrhenius ionic conductivity curve of amorphous L1.25NTCl within the temperature range of -60 to 70 °C. g SEM images of L1.25NTCl, Li6PS5Cl, and Li2ZrCl6 solid-state electrolytes. h 3D volume-rendered image generated by CT scanning, where the blue areas represent the voids within the solid-state electrolytes. i The porosity of L1.25NTCl, Li6PS5Cl, and Li2ZrCl6 solid-state electrolytes obtained through CT analysis.
Figure 4: Design and performance of ASSB suitable for operation in extremely cold conditions.

Cycling performance of ASSB at -30°C with charge/discharge potential range of 3.0-4.2 V vs. Li/Li and charge/discharge current density of 14 mA g-1. b Discharge curves of ASSB batteries at 18 mA g-¹ and different temperatures (-10, -30, and -40°C). c Cycling performance of ASSB batteries at -30°C at different multiplicities (18, 36, 54, 90, and 180 mA g-1 ) with potential range of 3.0-4.5 V vs. Li/Li. The cycling performance of ASSB cells at -30°C at 18 mA g-¹. The cycling performance of ASSB batteries was tested at -30°C with a current density of 18 mA g-¹ at a charge/discharge potential range of 3.0-4.5 V vs. Li/Li. e Charge/discharge curves of ASSB batteries at -30°C with a charge/discharge current of 18 mA g-¹ and with the cut-off potentials set at 3.0 V and 4.5 V vs. Li/Li, respectively. f -h Charge/discharge curves of ASSB at -60°C with cutoff potentials set to 3.0 V and 4.5 V vs. Li/Li electrodes with charge/discharge currents of 7.2 mA g-1 (f) and 18 mA g-1 (g, h), respectively. i Photographs of the ASSB operating at -60°C. All potentials relative to Li/Li were obtained by adding 0.6 V to the actual cell voltage, and all ASSBs were tested at a stacking pressure of 0.5 tons.
PART.3 Conclusion
Based on this study, we have successfully developed a cell using a novel amorphous solid state electrolyte (SSE) xLi₃N-TaCl₅ (optimal composition of L₁. ₂₅NTCI) all-solid-state battery (ASSB), which exhibits excellent electrochemical performance in extremely low-temperature environments (down to -60°C). By combining LiCoO₂ anode, L₁. ₂₅NTCI SSE, Li₁₀GeP₂S₁₂ (LGPS) interfacial layer, and Li-In anode in an optimized design, the ASSB achieves an initial discharge capacity of 183.19, 164.8, and 143.78 mAh g at -10°C, -30°C, and -40°C, respectively. -¹ initial discharge capacity (18 mA g-¹) and maintained 137.6 mAh g-¹ capacity after 100 cycles at -30°C (83.50% capacity retention). In addition, the battery was stably cycled for more than 200 hours at a current density of 18 mA g-¹ at -60°C, with a first discharge capacity of 51.94 mAh g-¹, verifying its utility and reliability under extreme cold conditions.

